Amino Acids (Basic, Acidic), Transamination & Proteins
Feb 17, 2023

Proteins are the most abundant macromolecules in living systems, and they play a crucial role in many biological processes. Similarly, amino acids are the building blocks of proteins, and their properties and interactions determine the structure and function of proteins. Therefore, understanding the structure and function of proteins and amino acids is essential for understanding many aspects of biochemistry.
Moreover, Amino acids and proteins are fundamental concepts in biochemistry, and they are likely to be tested in the NEET PG exam.
Read this blog further and learn more about this important biochemistry topic for NEET PG exam preparation.
Basic AA
- Arginine, Lysine and Histidine
- All are positively charged so all are Polar
- Comparison of Polarity
- Arginine (+++) > Lysine (++) > Histidine (+)
- Essential amino acid category
- Arginine (semi-essential)
- Lysine (essential)
- Histidine (semi-essential)
Acidic AA
- Aspartate, Asparagine, Glutamate and Glutamine
- all are negatively charged so all are Polar
- All are Non-essential AA Because all of them can be made from TCA intermediate as
- OAA → Aspartate → Asparagine
- α-KG → Glutamate → Glutamine
OH-Containing amino acid
| Serine | Threonine | Tyrosine |
| Non-essential | Essential | Non-essential |
| Polar | Polar | Polar |
Important Information
- Which AA has max. tendency to bind phosphate?
- Which AA is site for covalent modification?
- Which AA is involved in O–Glycosidic bonds?
Answer for all of these 3 questions is OH containing AA.
- So, in option choose option of OH containing AA.
- If 2 or more OH containing AA are given, SERINE is the best option to mark.
4. Which AA is involved in N–Glycosidic bonds?
Ans: Asparagine (has CONH which can provide N for N-glycosidic bond)
Sulfur Containing Amino Acids
1. Methionine
- Sulfur is attached to 2 carbons with strong bond (C–S–C)
- Non-polar (No -SH group)
- Essential AA
- Tip: difficult to attach Sulphur with 2 C
2. Cysteine
- Sulfur is attached to 1 carbon (C–SH)
- Polar (due to -SH group)
- Non-essential (can be made from methionine)
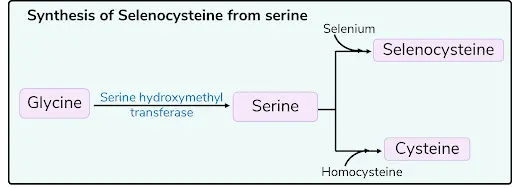
- First methionine is converted to homocysteine
- Homocysteine is converted to cysteine using serine and vit B6
Amino Acid
- Proline
- NH2 group is not Free (2o amine)
- Amino acid is attached to side chain forming a pyrrolidine ring
- Non-polar
- Non-essential (can be synthesized from ornithine in body)

Selenocysteine and Pyrrolysine
- 21st AA → Selenocysteine → Given by UGA
- 22nd AA → Pyrrolysine → Given by UAG
- UGA and UAG are stop codons and usually do not code for aa
- Exceptions: Selenocysteine & Pyrrolysine
- These AA are formed by Co–translational (during translation) modification (not by Post-translational modification)
Derived amino acids
- Def: AA not coming from codons but obtained from other AAs
- Two types
- Found in proteins
- Hydroxyproline, Hydroxylysine
- Not found in proteins
- present as free amino acids e.g. Ornithine Citrulline (Involved in Urea cycle)
- Homocysteine (Involved in methionine metabolism)
- Found in proteins
TRANSAMINATION
- 1st reaction in the catabolism of AA
- Reversible
- Requires B6 /PLP (pyridoxal phosphate)
- Common AA involved: Glutamate
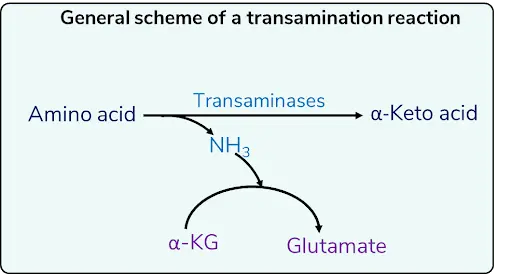
- Transaminases e.g. SGOT and SGPT
- only α-amino group can take part in transamination
- Exception is δ(delta) amino group of ornithine

- 17 amino acids can take part in transamination
- Rest 3 Amino acids can't take part in transamination e.g. proline (OH-proline), Lysine and Threonine
- 3 Amino acids that can't take part in transamination: Mnemonic: POLYTHENE
- PO - PrOline
- LY - LYsine
- THENE - THrEoNinE
Important Information
Transport form of NH3?
- From body and brain – Glutamine
- From Muscles - Alanine (via Cahill cycle)
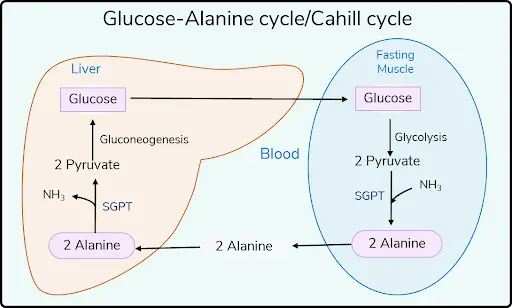
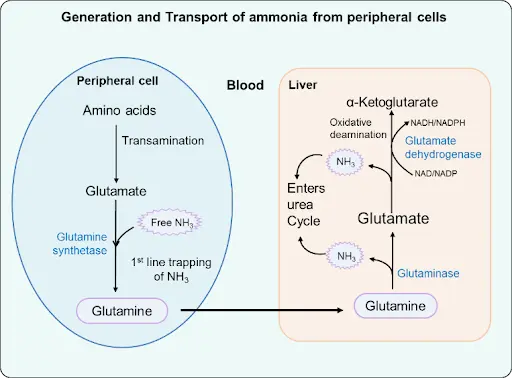
Transdeamination: Transamination (peripheral cells) + Oxidative deamination (liver)
Proteins
- Protein: polymers of amino acid
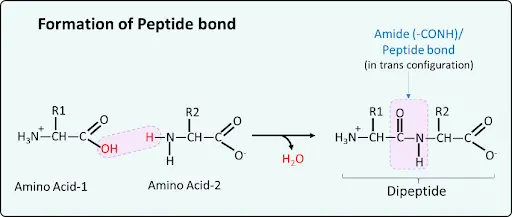
Amide Bond Formation
- Formed by removal of H2O when two amino acid joined together
- During this, carboxy gp of one amino acid and amine group of another amino acid reacts to form amide bond (-CONH-).
- If this is present in a protein it is called peptide bond.
- It is a strong covalent bond.
- It has partial double bond character which is in trans-configuration
Note: In fats, double bond is present but in cis-configuration
Structures of Proteins
Name Definition 1ᵒ Structure Sequence of amino acids 2ᵒ structure Obtained from folding of 1ᵒ structureα Helix (Symmetrical / helical structure)β Sheetsβ Turns 3ᵒ structure Further folding of 2o structure to form a fully folded 3ᵒ structure 4ᵒ structure > 1 polypeptide chain e.g. Hb (four polypeptide chains)
Monomeric protein: Those proteins which have only one monomer. They do not have Quaternary structure e.g. myoglobin
Important Information
AA not found in α-helix
- Proline and glycine create ‘bend’ in α helix
- Tryptophan due to bulky side chain
- Aspartate, Glutamate and Valine
|
Features |
1° |
2° |
3° |
4° |
|
Bond |
Covalent/ Peptide/ Amide |
Hydrogen bond |
|
(Mnemonic- HHI) |
|
Functional activity |
Absent |
Absent |
Present |
Present |
|
Denaturation |
Retained because peptide bond is very strong |
Lost |
Lost |
Lost |
|
Detection |
Mass spectrometry, Edman’s Technique |
|
||
Important Information
- Bonds in Enzyme-Substrate interactions (Mnemonic: HHI)
- H - Hydrophobic
- H - Hydrogen
- I - Ionic
- Sometimes covalent but Never Vander Waals Forces
- Bonds in Protein-DNA interactions (Mnemonic: HIV)
- H - Hydrophobic
- I - Ionic
- V - Vander Waals Forces
- Never Covalent bond
To study this topic in detail along with other important biochemistry topics, download the PrepLadder app and find high-quality video lectures and study material for NEET PG exam preparations.

PrepLadder Medical
Get access to all the essential resources required to ace your medical exam Preparation. Stay updated with the latest news and developments in the medical exam, improve your Medical Exam preparation, and turn your dreams into a reality!
Navigate Quickly
Selenocysteine and Pyrrolysine
Derived amino acids
TRANSAMINATION
Proteins
Amide Bond Formation
Structures of Proteins
Top searching words
The most popular search terms used by aspirants
- NEET PG Biochemistry
PrepLadder Version X for NEET PG
Avail 24-Hr Free Trial