Immunization and Vaccination in Children
Apr 28, 2023

Immunization In Children
When a person is immunized, usually through the administration of a vaccine, they become immune to or resistant to an infectious disease. Vaccines boost the body's natural defenses against sickness and infection by boosting immunity.
It is believed that vaccination prevents between 2 and 3 million deaths annually and is a proven method for eradicating infectious diseases that are life-threatening. It is one of the most economically beneficial investments in health, and it can be accessed by even the most vulnerable and difficult-to-reach people thanks to tested methods. It has well defined target populations, can be effectively administered through outreach initiatives, and vaccination doesn't call for a significant change in lifestyle.
In India free vaccines are provided against 11 life threatening diseases which includes
- Tuberculosis
- Diphtheria
- Pertussis
- Tetanus
- Polio
- Hepatitis B
- Pneumonia
- Haemophilus Influenzae type B (Hib)
- Measles
- Rubella
- Japanese Encephalitis (JE)
- Rotavirus diarrhea. (Rubella, JE and Rotavirus vaccine are available in selected states and districts)
National Immunization Schedule
Immunization is a fundamental requirement for all children as it is one of the most important and cost effective strategies for the prevention of childhood illnesses and disabilities and is thus a basic need for all children. And through this programme many childrens were helped which were not vaccinated because they were living in backward areas. Vaccine schedule of children is as explained in the following table.
|
Vaccine |
When to give |
Maximum age |
Dose |
Route |
Site |
|
For Pregnant Women |
|||||
|
Tetanus & adult Diphtheria (Td -1) |
early pregnancy |
-- |
0.5ml |
intramuscular |
Upper arm |
|
Tetanus & adult Diphtheria (Td -2) |
4 weeks after 1st dose of Td* |
-- |
0.5ml |
intramuscular |
Upper arm |
|
Tetanus & adult Diphtheria (Td booster) |
received 2 doses of pregnancy in the last 3yrs |
-- |
0.5ml |
intramuscular |
Upper arm |
|
For Infants |
|||||
|
BCG (Bacillus Calmette Guerin) |
At birth or to be given within 1 year of age. |
At birth till one year |
0.1 ml (0.05 ml until 1 month age) |
Intradermal |
Left Upper Arm |
|
Hepatitis B - Birth dose |
At birth or within 24 hours |
At birth within 24 hours |
0.5 ml |
Intramuscular |
Antero-lateral side of mid-thigh |
|
Oral Polio Vaccine (OPV) -0 |
At birth or within first 15 days |
Within the first 15 days |
2 drops |
Oral |
Oral |
|
Oral Polio Vaccine (OPV) -1,2,3, |
6 weeks, 10 weeks & 14 weeks |
Till 5 years of age |
2 drops |
Oral |
Oral |
|
Inactivated Polio Vaccine (IPV) 1 & 2 |
6 weeks & 14 weeks |
1 year of age |
0.1 ml |
Intradermal |
Right Upper arm |
|
Pentavalent vaccine (Diphtheria, Pertussis, Tetanus, Hepatitis B, Hib)- 1, 2 & 3 |
6 weeks, 10 weeks & 14 weeks |
1 year of age |
0.5 ml |
Intramuscular |
Antero-lateral side of mid-thigh |
|
Rotavirus Vaccine (RVV) 1, 2 & 3 |
At 6 weeks-1st dose 10 weeks-2nd dose 14 weeks-3rd dose |
1 year of age |
5 drops (lyophilized vaccine) |
Oral |
Oral |
|
Pneumococcal Vaccine (PCV) 1, 2 & Booster |
At 6 weeks-1st dose 14 weeks -2nd dose 9 months-booster |
1 year of age |
0.5 ml |
Intramuscular |
Antero-lateral side of mid-thigh |
|
Measles-Rubella (MR) 1 |
9 completed months - to 12 months. Given up to 5 yrs of age and if not received then at 9 - 12 months age |
5 years of age |
0.5 ml |
Subcutaneous |
Right upper arm |
|
Vitamin A (1st dose) |
At 9 completed months |
5 years of age |
1ml (1 lakh IU) |
Oral |
Oral |
|
Japanese Encephalitis (1st Dose)*** |
At 9 completed months - 12 months |
15 years of age |
0.5 ml |
Subcutaneous (live vaccine) Intramuscular (killed) |
Left upper arm and at the anterolateral side of mid thigh |
|
For Children and adolescents |
|||||
|
Diphtheria Pertussis Tetanus (DPT) booster 1 |
16- 24 months |
7 years of age |
0. 5 ml |
Intra- muscular |
Antero-lateral side of mid-thigh |
|
MR 2 |
16-24 months |
5 years of age |
0.5 ml |
Subcutaneous |
Right Upper arm |
|
OPV Booster |
16-24 months |
5 years of age |
2 drops |
Oral |
Oral |
|
Japanese Encephalitis***(if applicable) |
16-24 months |
15 years of age |
0.5 ml |
Subcutaneous |
Left Upper Arm |
|
Vitamin A***(2nd to 9th dose) |
18 months (2nd dose). After this one dose is repeated after every 6 months upto the age of 5 years. |
5 years of age |
2 ml (2 lakh IU) |
Oral |
Oral |
|
Diphtheria Pertussis Tetanus Booster (DPT) booster 2 |
5- 6 years |
7 years of age |
0.5 ml |
Intramuscular |
Upper arm |
|
Tetanus & adult Diphtheria |
10 years &16 years |
16 years of age |
0.5 ml |
Intramuscular |
Upper arm |
Pre-pregnancy doses of Td-2 or booster should be administered. But even if more than 36 weeks have gone, still provide these. If a woman in labor has never gotten Td, give it to her now.
JE vaccination at certain endemic districts; pentavalent vaccine (including diphtheria, pertussis, tetanus, hepatitis B, and hib)
As part of the Universal Immunization Programme, rotavirus vaccine (RVV)
Every year, 37 out of every 1000 children born in India do not live to see their fifth birthday, and diarrheal fatalities are one of the main causes of this. Of all the causes of diarrhea, rotavirus is the most common in children under the age of five. Rotavirus is thought to be responsible for 8,72,000 hospital admissions, 32,70,000 outpatient visits, and an estimated 78,000 fatalities per year in India. The availability of the Rotavirus vaccine will make it possible to tackle the issue of diarrheal deaths head-on.
A staggered rollout of the rotavirus vaccination began in 2016 with the original targeting of 4 states, and by the end of the year, 11 states had received it.
Pentavalent vaccinations
The DPT (diphtheria, pertussis/whooping cough, and tetanus), Hepatitis B, and Hib vaccines are included in the pentavalent vaccine. The immunization programme already includes the DPT and Hepatitis B vaccines. In the nation, pentavalent vaccines are gradually replacing them. Each intramuscular 0.5 ml dose of the pentavalent vaccine will be administered using syringes in the mid-thigh area.
When pentavalent vaccines are released, the updated immunization schedule will be as follows.
| Vaccine | Schedule |
| BCG, Hep B birth dose, OPV-O | At Birth |
| Pentavalent (which includes DPT + Hep B + Hib), OPV | 6 weeks, 10 weeks and 14 weeks |
| Measles and Vitamin A | 9-12 months |
| DPT booster, OPV booster, Measles2* | 16-24 months |
| DPT booster | 5-6 years |
Recommendations of IAP (Indian Academy of Pediatrics)
The national schedule has the full backing and endorsement of the Indian Academy of Paediatrics, the largest professional organisation of paediatricians in our nation. It offers the above-described regimen in addition to extra shots like the MMR (measles, mumps, and rubella) vaccination and the hepatitis B vaccine. It is important to keep in mind that although rubella may seem like a little disease, it has the serious potential to result in congenital problems in a baby whose mother is not immune to the infection and contracts it during the first trimester.
| Age | Vaccines |
| Birth | BCG, OPV 0, Hepatitis B -1 |
| 6 weeks | OPV-1, Penta-1, fractional-IPV-1, PCV-1, Rota-1 |
| 10 weeks | OPV-2, Penta-2, Rota-2 |
| 14 weeks | OPV-3, Penta-3, Rota-3, Fractional IPV-2, PCV-2 |
| 6 months | Influenza 1 |
| 7 months | Influenza 2 |
| 6-9 months | Typhoid Conjugate Vaccine |
| 9 months | MMR I, PCV-booster, Japnese encephalitis, Vita-A-1 |
| 16-24 months | DPT Booster-1, Measles Rubella -2, OPV Booster, Japnese Encephalitis-2, Vit-A-2 |
| 5-6 Years | DPT-Booster 2 (No OPV) |
| 10-16 Years | TD (Instead of TT) |
Abbreviations of the Vaccination Are
- BCG: Bacillus Calmette Guerin
- OPV: Oral poliovirus vaccine
- DTwP: Diphtheria, tetanus, whole cell Pertussis
- DT: Diphtheria and tetanus toxoids
- TT: Tetanus toxoid
- Hep B: Hepatitis B vaccine
- MMR: Measles, Mumps and Rubella Vaccine
- Hib: Haemophilus influenzae Type ‘b’ Vaccine
- IPV: Inactivated poliovirus vaccine
- Td: Tetanus, reduced dose diphtheria toxoid
- HPV: Human Papillomavirus Vaccine
- PCV: Pneumococcal Conjugate Vaccine
- TdaP: Tetanus and Diphtheria Toxoids and a Cellular Pertussis Vaccine
Mission Indra Dhanush
- It was Launched by the Ministry of health and family welfare by the Government of India on 25th Dec 2017.
- Mission was to Improve immunization Courage from 65%, in 2014 at least 96 % over 5 years through special catch up drives.
- It Included vaccines against
| TB (BCG) | Polio |
| Dip | Measles |
| Pertesis | Hep B |
| Tetanus | - |
Queries Related To Vaccination
- If a child is +snt with upper respiratory tract infection on the due date of vaccination then Give vaccines to this child there is no need to postpone the vaccinations in case of minor diseases. Vaccine to be postponed only During serious illness.
- If vomiting occurs after OPV then in this case If vomiting had occurred within 30 minutes then repeat the vaccination otherwise not.
- In case of Lapsed (missed vaccination on the due date) immunization: We will continue only from missed vaccines there is no need to restart vaccine series regardless of time. Give the vaccine as per schedule.
- Preponed immunization: If given 5 or more days before the due date then that dose is not counted we need to repeat it.
- Unknown immunization status: Child should be considered immunized and vaccinated accordingly.
- What vaccines should be given to unimmunized child: measles Rubella Vaccine
- DTP (7 year – Tdap)
- Hib (If < 5 years age)
- OPV (If < 5 year age)
- BCG (if <1 year age)
- -Hep B (if 1 year age)
- Vaccines contraindicated in Egg allergy_ Yellow fever & Influenza
- Vaccines causing Thrombocytopenia – Measles vaccines
- Strains including in meningococcal vaccine – A, C, Y w – 135
- Streat sensitine vaccine – OPV, re. cons t. BCG, Measles
- Light sensitive vaccine – Measles, BCG, Rota V, Jap E
- Freeze sensitive vaccine – Hep B, Penta valent, TT, DPT
Vaccine vial monitor
- Heat sensitive labels are present on the vial that indicate cumulative heat exposure over time to that vaccine vial.
- If color of square is lighter than the outside circle → the we can use vaccine
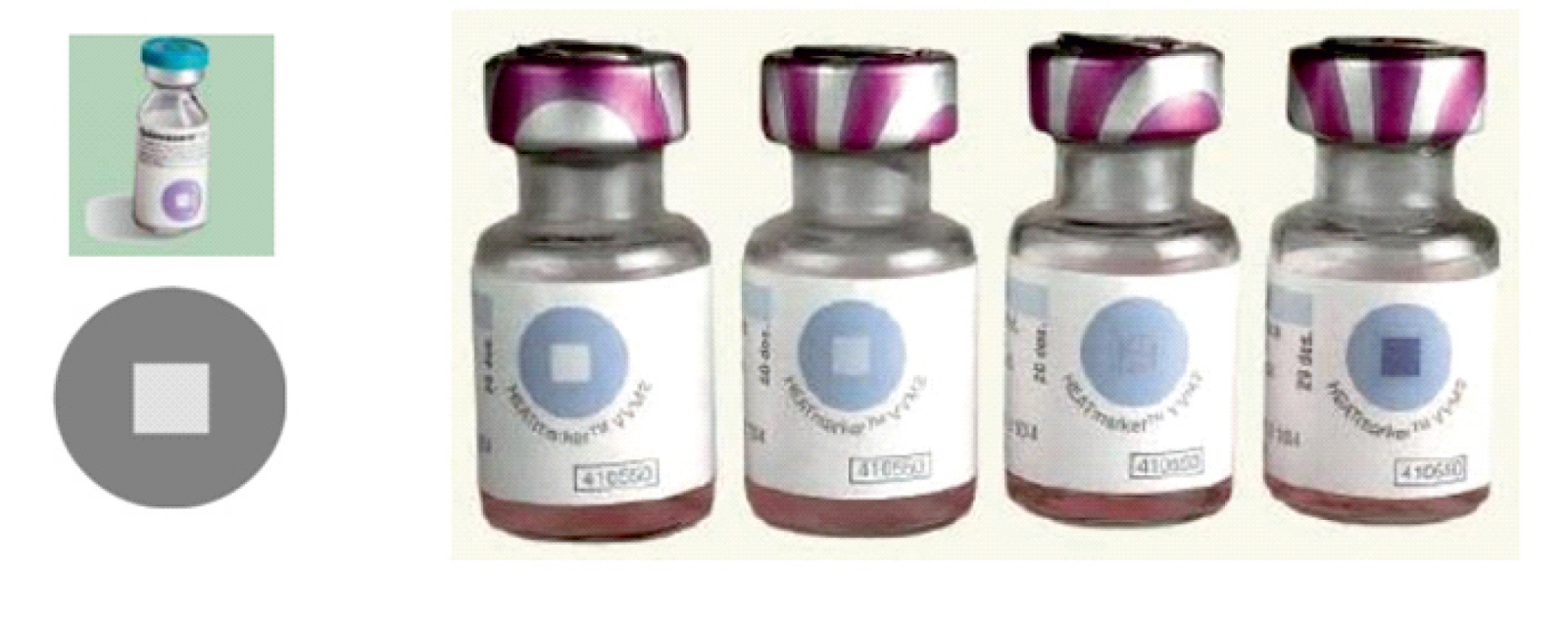
- If color is same or darker →then we can't use vaccine
- Number present on the vial Indicates the no. of Days, the vaccines Remain potent, when exposed to 37°C.
- Test done to check for cold Damage to the vaccine due to free tiny is k/n as ‘SHAKE TEST. In this test the vaccine vial is shaked and if the Precipitate is formed then this vaccination vial should be disposed of.
Open VIAL Policy
It allows the reuse of partially used multidose vials in subsequent immunization sessions (upto 4 weeks) in following conditions
- Expiry date not reached
- Cold chain is maintained
- Date of opening of vial is clearly mentioned
- Aseptic technique used to withdraw vaccine
- Vaccine vial septum not submerged in water
Open vial policy is applicable for the following vaccines:
- DPT
- TT
- Hep B
- PCV
- Pentavalent
- OPV
- IPV
Open vial policy is not applicable for following vaccines:
- Measles/MR
- BCG
- Rota virus
- JE vaccine
Vaccines Recommended in Adolescent
- Tdap – HPV
- Td – Influenza Rabies
- TT – Japanese encephalitis
- Pneumonococcal
Immunization in special situations
Immunization in Special Situations
- In HIV positive children
- In asymptomatic HIV positive children, all vaccines can be given except OPV
- In symptomatic children with HIV, all live vaccines are contraindicated while all killed vaccines can be given
- In Immunocompromised child (including child on oral steroids/chemotherapy)
- These children should not receive any live vaccine till at least 1 month after discontinuation of steroids or chemotherapy
- It can cause that disease (against which vaccination is given) due to low immunity
- Killed vaccine can be safely given (immunogenicity might be doubtful)
- Pneumococcal vaccines and annual influenza vaccines must be given
- Sibling of these children should not receive transmissible live vaccine like OPV
- Oral polio virus is going to be shed in stools and these children with household contact can get infected
- These children should not receive any live vaccine till at least 1 month after discontinuation of steroids or chemotherapy
- For a child on corticosteroids, Immunization schedule:
- Needs to be modified if - Child is on oral steroids at a dose of ≥ 2 mg/kg/day or receiving ≥ 20 mg/day prednisolone
- Does not need to be modified if – Child is on inhaled corticosteroids (bronchial asthma) or topical steroids or ointment or cream (dermatological condition)
- Immunization of a child with
- Primary immunodeficiency
- Planned splenectomy
- Certain conditions like hereditary spherocytosis, hypersplenism, thalassemia or Gaucher disease with massive spleen and hypersplenism, refractory ITP
- In all children with severe immunodeficiency whether it is B cell group i.e., X-linked agammaglobulinemia or severe T cell immune deficiency like SCID (Severe Combined Immunodeficiency) – All live vaccines are contraindicated
- Ensure at least 2 weeks before a planned splenectomy, child should have been immunized against capsulated organisms like pneumococcus, meningococcus & Haemophilus influenzae (As spleen protects against these capsulated organism)
- For pneumococcal vaccine – PCV (give immune memory) + Polysaccharide vaccine should be given (protects against all 23 strains of pneumococcus)
- Immunization of child with known coagulation disorder (e.g., Hemophilia)
- There is formation of large bruises on site of intramuscular injection and sometime bleeding may not stop after injection
- Give vaccines subcutaneously instead of intramuscularly so chance of formation of hematoma is less e.g., Hib, Pneumococcal polysaccharide vaccines
- Schedule immunization, shortly after administration of factor therapy (factor 8 in case of hemophilia)
- Use smaller size needle (23 G or even smaller than that)
- Apply firm pressure at injection site for at least 5-10 minutes as to stop bleeding quickly
- Immunization of babies born preterm
- All vaccines can be given at chronological age as recommended by National Immunization Schedule
- Birth vaccines – BCG and OPV-0 are given after initial stabilization (preferably at time of discharge)
- So other vaccines will also be delayed by 4 weeks according to general principle of immunization
Hep B is given as soon as possible after birth to prevent vertical transmission from mother to child along with Vit k.
BCG (Bacillus Calmette Guerin) vaccine
Calmette and Guerin were the first scientists who invented BCG vaccine Against bacteria: Mycobacterium tuberculosis Strain commonly used: Copenhagen (Danish 1331) or Pasteur strain. 3Ls
- Live attenuated vaccine
- Light sensitive (comes in dark ampoule)
- Lyophilised vaccine (comes in powder form and needs to be dilute with diluent)
The Diluent used for BCG vaccine Normal saline is used as diluent (supplied along with vaccine vial) Distilled water is not used as it will irritate the skin. The Dosage is 0.05 ml till 1 month of age and Beyond 1 month: 0.1 ml. Route of administration: Intradermal. Maximum age till BCG can be given as per NIS guidelines: 1 year. In India as tuberculosis is endemic condition so by 1 year of age all the children would have been exposed to blood TB virus in some way or another IAP used to recommend BCG vaccine till 5 years of age but according to NIS it is to be given till 1 year of age .Normal response following BCG vaccination
Papule → Ulcerate → Crust formation → Scar formation (by age of 6-8 weeks)
- Adverse effects of bcg vaccine are:
- Suppurative lymphadenitis (common)
- Lymph nodes of left axilla becomes enlarged and become pus filled
- BCG osteitis (bone infection)
- Disseminated BCG infection (in immunodeficient child)
- Suppurative lymphadenitis (common)
- Protective effect: 0-80%
- It does not give protection against pulmonary COX much
- It protects mainly against severe forms of TB i.e., TB meningitis or disseminated TB
- It does not give protection against TB infection
Polio vaccines
There are 2 types of polio
- OPV (Oral polio vaccine)/Sabin: Live attenuated vaccine
- IPV (Injectable polio vaccine)/Salk: Killed vaccine
- Strains of polio virus used in OPV: P1 & P3 P2 strain was globally discontinued in April 2016. According to WHO recommendations P2 strain to be no longer used in OPV. Only P1 and P3 strains are used in OPV. WHO no longer recommends an OPV only schedule; at least 1 dose of IPV should be there in NIS. IPV not only protects against polio virus but it also protects against disease caused by vaccine derived polio viruses
VAPP (Vaccine Associated Paralytic Polio)
- Cases of AFP (Acute flaccid paralysis) having residual weakness 60 days after the onset of paralysis & from whose stool samples vaccine related polio viruses have been isolated
VDPV (Vaccine Derived Polio Virus)
- Arise due to mutation and recombination of the vaccine polio virus in human gut
- They are 1-15% divergent from parent vaccine strain
Hepatitis-B vaccine
Route of administration: Intramuscular, on anterolateral aspect of thigh. Dosage: Birth dose: As soon as possible after birth It helps in prevention of transmission of hepatitis B infection from mother to baby. Subsequent dosage is given as part of Pentavalent vaccine according to NIS at – 6,10,14 weeks. If the mother is Hepatitis B positive then Hep B vaccine is given immediately after birth to the baby. If Hep B negative immunoglobulin ideally within 12 hours after birth
Complete Hep B vaccination series at 2 & 6 months
DPT vaccine
DPT vaccine protects against 3 diseases i.e. Diphtheria, Pertussis, Tetanus. Dosage is given at 6 weeks, 10 weeks, 14 weeks. Booster: 16-24 weeks. 2nd booster: 5-6 years. It is given as a part of pentavalent vaccine according to NIS. Route of administration Intramuscular, on anterolateral aspect of left thigh. Adverse effects are:
- Local effects: Redness, pain at site of injection
- Systemic effects: Fever
- Severe adverse effects
- Persistent inconsolable cry (> 3 hours)
- Seizures
- Hypotonic hyporesponsive episodes
- Encephalopathy/altered sensorium
- Anaphylaxis
Contraindications to DPT vaccines
- Progressive neurological illness
- Cerebral palsy (static neurological illness): Not a contraindication to DPT vaccine
- Anaphylaxis to a previous dose of DPT vaccine
- Encephalopathy within 7 days of previous dose of DPT vaccine
Recommended catch up schedule for children who did not receive DPT vaccine
- ˂ 7 years of age: DPT is given at 0,1,6 months
- > 7 years of age: Tdap (lesser dose of Diphtheria and Pertussis) at first visit
- Followed by Td & Td: 0,1,6 months
Pentavalent vaccine
- Component of pentavalent vaccine used in National Immunization Schedule
- Diphtheria
- Pertussis
- Tetanus
- Hepatitis B
- H. influenzae type B
- Dosage, route, and site of administration: 0.5 ml intramuscularly at anterolateral aspect of left thigh at 6, 10, 14 weeks according to NIS
Measles vaccine
It is a Live-attenuated vaccine. Strain used for the measles vaccine are the Edmonston Zagreb stain. Diluent used for the measles vaccine is Distilled water. Route: Subcutaneous route (right upper arm). Recommended age: 9 completed months – 12 months of age. In case of measles outbreak: Measles vaccine can be given between 6-9 months of age as post exposure prophylaxis
- Dose given before 9 completed month age is not counted as part of NIS
- If a dose of measles vaccine is given before 9 months, it is still required to be given at 9 months and booster dose – 16-24 months
- Adverse effects
- Local effects: Pain, redness, tenderness
- Fever
- Mild measles like illness (rashes)
- ThrombocytopeniaPneumococcal vaccine
- There are 2 types of pneumococcal vaccine
|
PCV (Pneumococcal conjugate vaccine) |
PPV (Pneumococcal polysaccharide vaccine) |
|
|
|
|
|
|
|
|
- Advantage of PPV: It protects against 23 strains of pneumococcus
- It gives much wider protection than PCV
- Recommendation about pneumococcal vaccine in our National Immunization schedule
- PCV: 13: 6, 14 weeks & booster dose at 9 months
- Dose: 0.5 ml given at anterolateral aspect of right thigh
- Catch up immunization schedule – till 1 year of age
- Route of administration of both PCV & PPV: Intramuscular
Rotavirus vaccine
It is Recently introduced in the National Immunization Schedule. Dosage is given on 6, 10, 14 weeks. It is a Type of Live attenuated oral vaccine. Recently, 116 E strain of rotavirus was found in AIIMS NICU. It is now used to produce Indian rotavirus vaccine
- Adverse effects
- Intussusception
- Rotavirus vaccine is contraindicated if child has history of intussusception
- Catch up immunization schedule: Permissible upto 1 year of age
Hepatitis A Vaccine
It is not a part of the National Immunization Schedule. But the Indian Academy of Pediatrics recommends that it should be given to all healthy children. Type of vaccines
- Live vaccine: Single dose
- Age of administration for live vaccine: 12-23 months
- Inactivated vaccine: 2 doses required 6 months apart
- First dose is given at 1 year of age
Typhoid Vaccine
- Types of typhoid vaccine are:
- Vi Capsular polysaccharide vaccine (Used previously)
- Typhoid Conjugate vaccine (newer vaccine)
Typhoid VI Capsular polysaccharide vaccine
- It can be given only beyond 2 years of age
- No good immune memory → Repeat dose is required every 3 years
- Efficacy: 50-60%
Typhoid conjugate vaccine
- It can be given at ≥ 6 months of age
- Single dose is given
- Efficacy: 90%
Varicella Vaccine
- It is a type of Live-attenuated vaccine
- Strain used for varicella vaccine is Oka strain
- Route of administration: Subcutaneous
- Dosage
- 1st dose: 15 months of age
- 2nd dose: 4-6 years of age
- IAP recommendations
- It should be given to all children with no previous history of varicella
- For post- exposure prophylaxis
- Ideally given within 3 days of exposure, but can be given upto 5 days following exposure to prevent development of varicella infection
HPV (Human Papillomavirus) vaccine
- It is a recombinant DNA vaccine
- Types
- Quadrivalent vaccine (Gardasil)
- It protects against HPV serotype 6,11,16,18
- Bivalent vaccine (Cervarix)
- It protects against HPV serotypes: 16,18
- Quadrivalent vaccine (Gardasil)
- Protective efficacy: 99% against vaccine type related genital warts & vulvar and vaginal neoplasia
- Minimum age at which HPV vaccine recommended: 9 years
- It should be started before first sexual activity
- HPV should be given to all female children
- Maximum age for catch up schedule: 13-45 years of age
- Recommendation according to WHO SAGE (Strategic Advisory Group of Expert/ Committee) protocol for HPV vaccination (April 2022)
- For 14-19 years old girls - Either 1 or 2 doses of HPV vaccine can be given
Influenza Vaccine
- Types of vaccine of influenza includes:
- Inactivated vaccine: Whole virus/Split product
- Egg based vaccine
- Cell culture vaccine (for egg allergic individuals)
- Recombinant technology derived vaccines
- Nasal live attenuated vaccine (not available in India)
- Regime for inactivated vaccine: 2 doses intramuscular, 4 weeks apart followed by annually
- Every year strains of Influenza vaccine changes based on WHO recommendation
- Annual influenza vaccine should be given to all the high risk individual like elderly immunocompromised children, or children with chronic disease
- Latest WHO recommendation for the Influenza vaccine composition for 2022-23 for northern hemisphere
- A/Wisconsin/588/2019 (H1N1)
- A/Darwin/6/2021 (H3N2)
- B/Austria/1359417/2021 (B/Victoria Lineage)
- B/Phuket/3073/2013 (B/Yamagata lineage)
This is everything that you need to know about IMMUNIZATION for your PEDIATRICS PREPARATION. For more interesting and informative blog posts like this download the PrepLadder App and keep reading our blog!

PrepLadder Medical
Get access to all the essential resources required to ace your medical exam Preparation. Stay updated with the latest news and developments in the medical exam, improve your Medical Exam preparation, and turn your dreams into a reality!
Navigate Quickly
Immunization In Children
National Immunization Schedule
Pentavalent vaccinations
Recommendations of IAP (Indian Academy of Pediatrics)
Abbreviations of the Vaccination Are
Mission Indra Dhanush
Queries Related To Vaccination
Vaccine vial monitor
Open VIAL Policy
Open vial policy is applicable for the following vaccines:
Open vial policy is not applicable for following vaccines:
Vaccines Recommended in Adolescent
Cocoon Strategy
Immunization in special situations
BCG (Bacillus Calmette Guerin) vaccine
Polio vaccines
VAPP (Vaccine Associated Paralytic Polio)
VDPV (Vaccine Derived Polio Virus)
Hepatitis-B vaccine
DPT vaccine
Pentavalent vaccine
Measles vaccine
Rotavirus vaccine
Hepatitis A Vaccine
Typhoid Vaccine
Varicella Vaccine
HPV (Human Papillomavirus) vaccine
Influenza Vaccine
Top searching words
The most popular search terms used by aspirants
- neet pg pediatrics
PrepLadder Version X for NEET PG
Avail 24-Hr Free Trial

